
30 Mar The STAR Project & Spearheading the Data Modernization Effort
Background and Challenges:
In 2018, Congress passed the legislation, the Childhood Cancer Survivorship, Treatment, Access and Research (STAR) Act, with the goal of advancing understanding and care of cancer diagnoses in children, adolescents and young adults. This legislation empowered the CDC’s National Program of Cancer Registries (NPCR) to build the infrastructure required for rapid case reporting from facilities in which cancers are diagnosed and reported to state central cancer registries (CCRs). More timely and efficient reporting leads to improved data availability, helping to better understand cancer in young people and connect those affected to clinical trials, resources, and support networks. To help accomplish the STAR Act’s goals, NPCR contracted with Tanaq Support Services (Tanaq) to work directly with state cancer registries and laboratories. Tanaq’s goals included the following: expand rapid case reporting from facilities where cancer is diagnosed into cancer registries; develop and implement a technological infrastructure to facilitate rapid reporting processes and provide timely data to improve understanding and the lives of children, adolescents and young adults diagnosed with cancer.
Tanaq’s Approach:
The Right Experts Leading the Charge:
Tanaq, under the guidance of an experienced project manager, assembled three specialized teams—functional, technical, and communication—each equipped with distinct expertise and skills tailored to effectively accomplish the objectives of the STAR Project.
Recruitment of States & Alignment with State’s Needs:
The functional team crafted a selection matrix to identify the best partner states for the project. Ohio, Georgia, Nebraska, and Rhode Island were ultimately chosen, with each state having unique goals. Ohio and Nebraska hoped to expand their electronic pathology (ePath) reporting capabilities, Georgia wanted to support the development of a new technological infrastructure for reporting, and Rhode Island planned to address interstate data exchange process challenges. Throughout the project, the functional team ensured that they aligned the states’ resources and desires with the project goals and addressed questions and challenges as they arose. This included engagement tracking and the development of metrics to assess the current state of rapid reporting within the registry.
Within each state, the functional team actively engaged and recruited entities that report childhood, adolescent, and young adult cancer cases, as well as encouraged them to pilot the National Program of Cancer Registries National Oncology Rapid Ascertainment Hub (NPCR-NOAH), a cloud-based infrastructure developed by the technical team to optimize pathology reporting. The team contacted leaders at 19 central registries and 28 facilities, held information-gathering meetings and conducted an analysis of fit and feasibility using an assessment checklist. Following these steps, 3 central registries and 5 facilities took part in the pilot study. As the pilot progressed, the functional team monitored each state’s successes and regularly hosted meetings for participating states to come together, provide feedback and exchange ideas for best implementation strategies.
Building New Technology and Improving Existing Infrastructure:
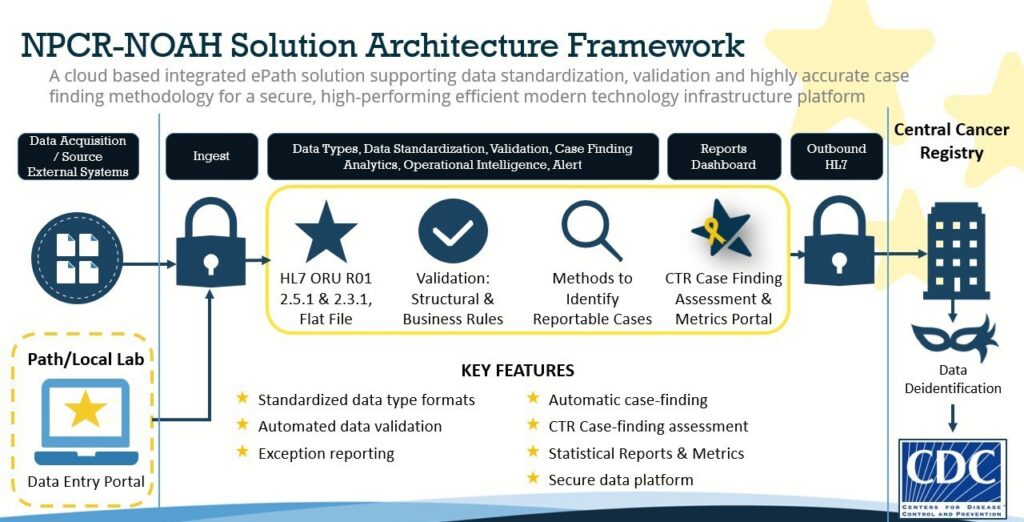
As a cornerstone of CDC’s larger Data Modernization Initiative (DMI), Tanaq and CDC developed NPCR-NOAH to help state registries receive and rapidly classify reportable cancers and process data faster.
NPCR-NOAH is an adaptable, cloud-based infrastructure that supports early inclusion of childhood cancer cases into state cancer registries. An informatics system designed to improve case finding and timeliness of reporting pediatric, adolescent and young adult cancer cases. (Improving case findings is crucial because many laboratory reports are submitted in narrative form, lacking discrete fields, making it difficult to determine if they represent reportable cancer cases.) Security is paramount since Personally Identifiable Information (PII) is processed. As such, several layers of security were implemented including compliance with Federal Authority to Operate requirements, NIST 800, data encryption and Multi-Factor Authentication.
The technical team ensured that states could submit pathology reports to CCRs using HL7 and standardized flat file formats to track the epidemiology of childhood and young adult cancer cases. They also developed and tested a tool called the NPCR-NOAH Certified Tumor Registrar (CTR) Observation Portal which allowed CTRs access to their state’s data and enabled them to review lab reports and assess their reportability. The team used results of the CTR analysis to make improvements to NPCR-NOAH’s case finding algorithm. Additionally, the team created a Metrics Dashboard to evaluate statistics on system performance and case finding accuracy.
Because each state had different resources and processes, the team provided flexibility with implementation that complemented the existing registry infrastructure/processes, with the goal of making it faster without sacrificing quality.
Providing Central Registry Operational Support:
Certified Tumor Registrars (CTRs) came onboard to help states with the following tasks: providing abstracting activities for pediatric, child, and young adult cancer cases; following up with reporting facilities when cases were not reported within 30 days or when they were incomplete; resolving and reporting issues discovered through auditing NPCR-NOAH; and developing job aids to assist reporting facilities with rapid case reporting.
A Layered Communication Strategy:
From parents, cancer researchers, physicians, the surveillance community, legislators, politicians, advocacy, and support groups…, unique populations take interest in the STAR Project for diverse reasons. To address this, the communication team constructed a message matrix to craft tailored, targeted content, created listservs, and developed standard operating procedures to streamline communication deliverables. The team generated various types of content, such as PowerPoint presentations for meetings and conferences, fact sheets, web content, abstracts, and poster presentations. Additionally, they produced the quarterly “Shooting Star” newsletter, featuring summary articles on new cancer studies, interviews, and updates on the STAR Project.
Project leadership expressed a specific desire to expand human interest stories from folks personally impacted by cancer. To accomplish this, the communication team researched and reached out to potential interviewees and conducted both compelling email and video interviews with those who agreed to participate. Their stories are powerful and put names and faces to the people we hope will benefit the most from the STAR Project endeavors.
Performance Benchmarks and Notable Accomplishments:
Each Participating State Reached Key Milestones:
Georgia successfully completed the NPCR-NOAH pilot study. 1,479 lab reports were selected from 22 facilities for inclusion. Without any changes to the case finding algorithm, NPCR-NOAH accurately classified reportability for 94.5% of cases.
Nebraska successfully completed the NPCR-NOAH pilot study. De-identified data and 261 lab reports from 5 independent labs were processed in NPCR-NOAH for 3 weeks. The pilot study successfully validated the following: connectivity and file transfer using NPCR-NOAH; we processed data into the Central Cancer Registry’s ePath system and main cancer registry system; we conducted a reportability assessment using the NPCR-NOAH’s CTR Observational Portal, and we generated and distributed summary reports daily. Without any changes to the case finding algorithm, NPCR-NOAH accurately classified reportability for 75% of cases. Nebraska also developed a de-identification data tool that the STAR Project team helped enhance and test to validate its functionality and accuracy.
Rhode Island’s Cancer Registry and one of its hospital facilities have been piloting NPCR-NOAH since August 2023. Before the pilot study, the hospital facility took 14 months to report a case. Since implementing NPCR-NOAH, overall reporting timeliness improved to 9 months.
Ohio and the STAR Project Team met with the state’s top 5 high-volume reporting facilities for patients in the 0-29 age group. The goal was to help these facilities improve their reporting of cases within a 6-month timeframe. Following these discussions, several facilities took steps to improve reporting timeliness and one facility agreed to pilot NPCR-NOAH. While Ohio has been steadily improving reporting timeliness for 5 years, it jumped 18% following these discussions.
Implementing the M Record: Hospitals and care facilities usually waited until a cancer case had finished treatment before reporting to the CCR. Since treatment could take weeks or even months/years, many cases were reported late. Ohio revised their reporting requirements to allow for reporting even without completed treatment and started accepting follow-up submissions containing treatment data (M record). After piloting with one PAYAC facility, the change was implemented across all reporting facilities.
First of a Kind:
NPCR-NOAH received the authorization to operate (ATO) for processing moderate PII, a vital step for the project’s longevity. This includes security certification, security accreditation, e-authentication, and business continuity planning. ATO was a big achievement as it was the 1st external cloud system at NPCR to receive an ATO.
The File Riddle: Overcoming Barriers in Electronic Reporting:
The North American Association of Central Cancer Registries’ (NAACCR) Workgroup (WG) for the Pathology Laboratory Electronic Reporting, Volume V updated the existing pipe-delimited flat file standard that had been in place since 2009. A pipe-delimited flat file is like a digital table where information is organized using vertical lines (pipes) to separate different pieces of data. It’s a simple way of storing data in a plain text file. The long-term goal is to have all laboratories report data using a Health Level Seven (HL7) format, following specifications defined by the NAACCR WG. HL7 is a set of international standards for the exchange, integration, sharing and retrieval of electronic health information that ensures interoperability between different software systems. Many reporting laboratories face challenges with using an HL7 format due to technological or resource constraints.
The STAR Project Team and CDC compared the existing NAACCR pipe delimited specification, NPCR-NOAH’s flat file specification, and the NAACCR Volume V HL7 specification. The team submitted recommendations on adjustments and implementation to the NAACCR Vol V Workgroup for additional review. Anticipated outcomes include the Workgroup’s approval of these recommendations, improved rapid reporting from laboratories, and broader acceptance of NPCR-NOAH for electronic pathology reporting.
Getting the Word Out:
The STAR Project Team presented two abstracts at the 2023 North American Association of Central Cancer Registries (NAACCR) Conference in New Orleans. One was a poster presentation showing how the adoption of a standardized template for early case reporting was feasible and its potential for improving reporting timeliness. Conference leaders awarded it first place in the Annual Conference Poster Presentation for Standards and Registry Operations. The second was a live slide presentation by the project team on the successes and barriers for state participation in the NPCR-NOAH pilot study:

The team also presented the abstracts at the 2024 National Cancer Registrars Association’s (NCRA) conference in Indianapolis and took third place in the poster presentations. Further, the team showcased the NPCR-NOAH CTR observational portal, a secure web-based application, at the NAACR 2004 conference, which allows subject matter experts to confirm the correct classification of pathology reports by NPCR-NOAH. Finally, the communication team sent four “Shooting Star” newsletters with open and reach metrics exceeding the industry standard.
Moving Forward:
The STAR Project pioneers the adoption of a cloud-computing system within NPCR, laying the foundation for implementing and scaling rapid electronic reporting. Its insights gleaned from sourcing and assessing potential electronic reporting avenues, alongside the development of adaptable technology tailored to state requirements, serve as a valuable blueprint for advancing public health data modernization initiatives.
Join Our Team
Are you inspired by the leadership and opportunities at Tanaq? Explore exciting career opportunities with us and become part of a team that values innovation, growth, and excellence. Visit our Careers Page today to learn more and apply!
We’d also love to connect with you in person—meet us at the upcoming National 8 (a) Small Business Conference in New Orleans.
Sorry, the comment form is closed at this time.